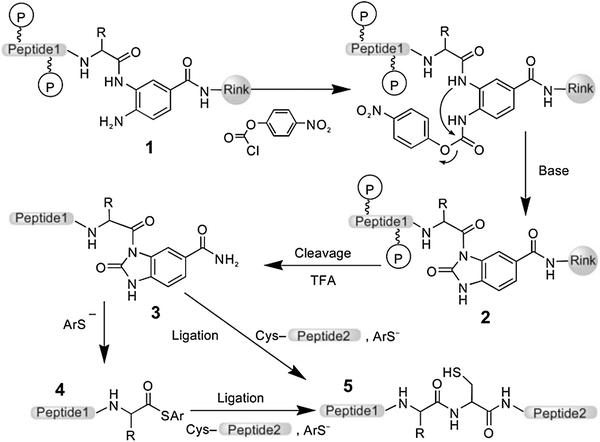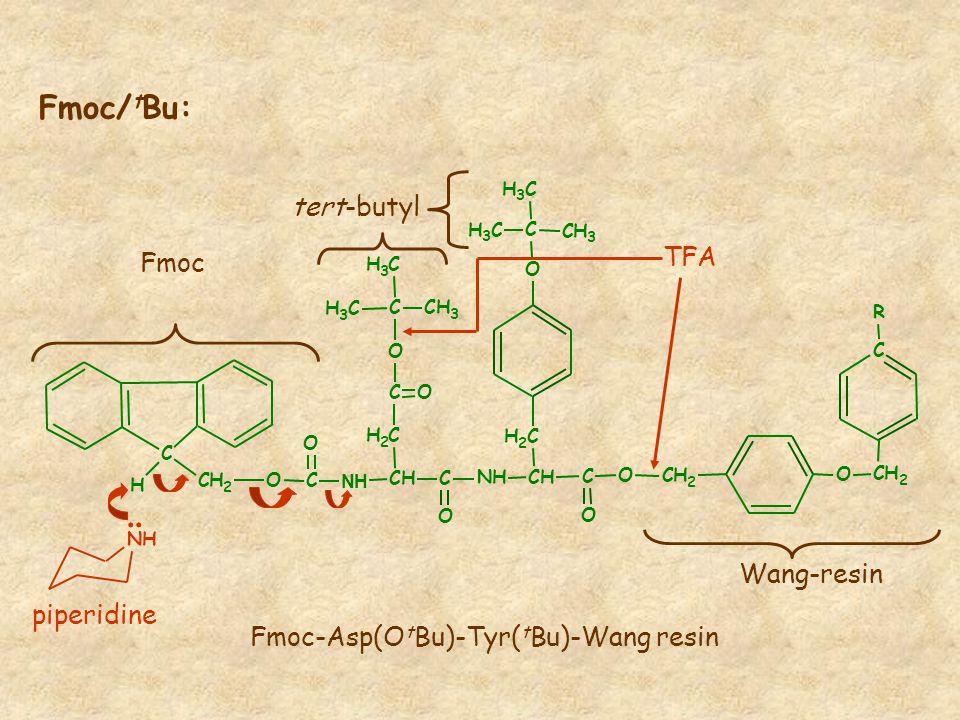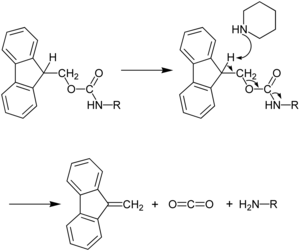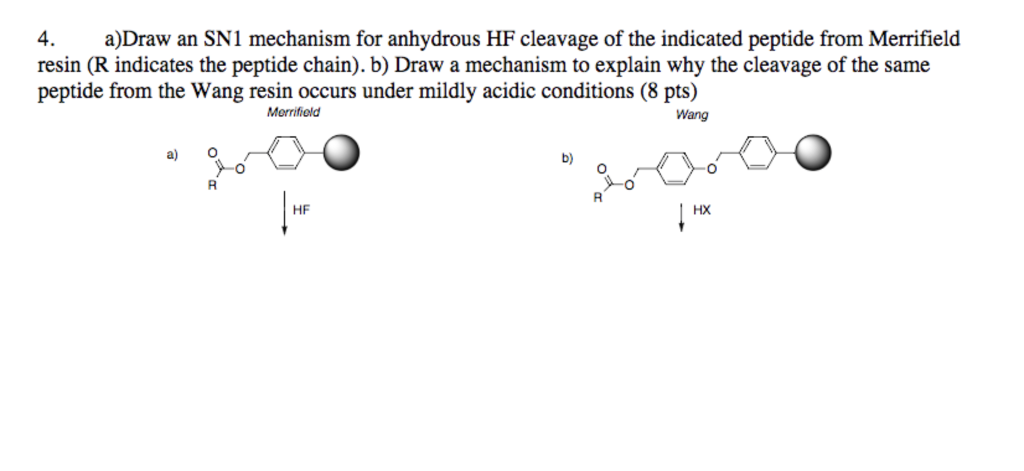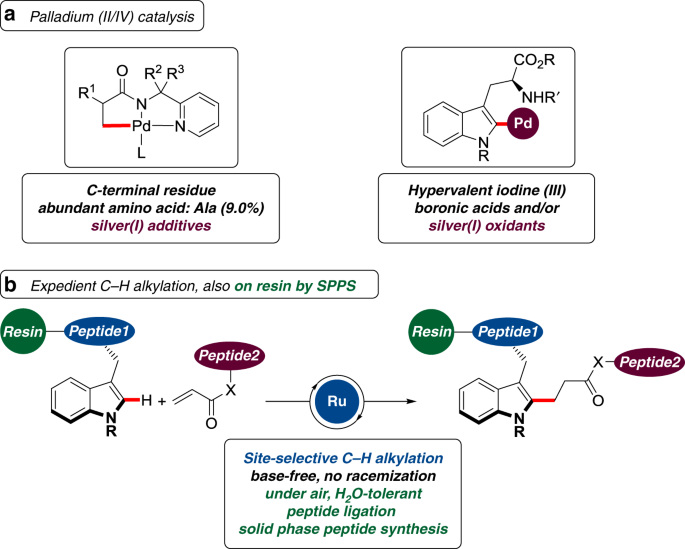
Late-stage peptide C–H alkylation for bioorthogonal C–H activation featuring solid phase peptide synthesis | Nature Communications
Conotoxin analogues, and methods for synthesis of intramolecular dicarba bridge containing peptides - Patent 1984384

Synthesis of Peptoids Containing Multiple Nhtrp and Ntrp Residues: A Comparative Study of Resin, Cleavage Conditions and Submonomer Protection. - Abstract - Europe PMC

Frontiers | Recent Progress in the Chemical Synthesis of Class II and S-Glycosylated Bacteriocins | Microbiology
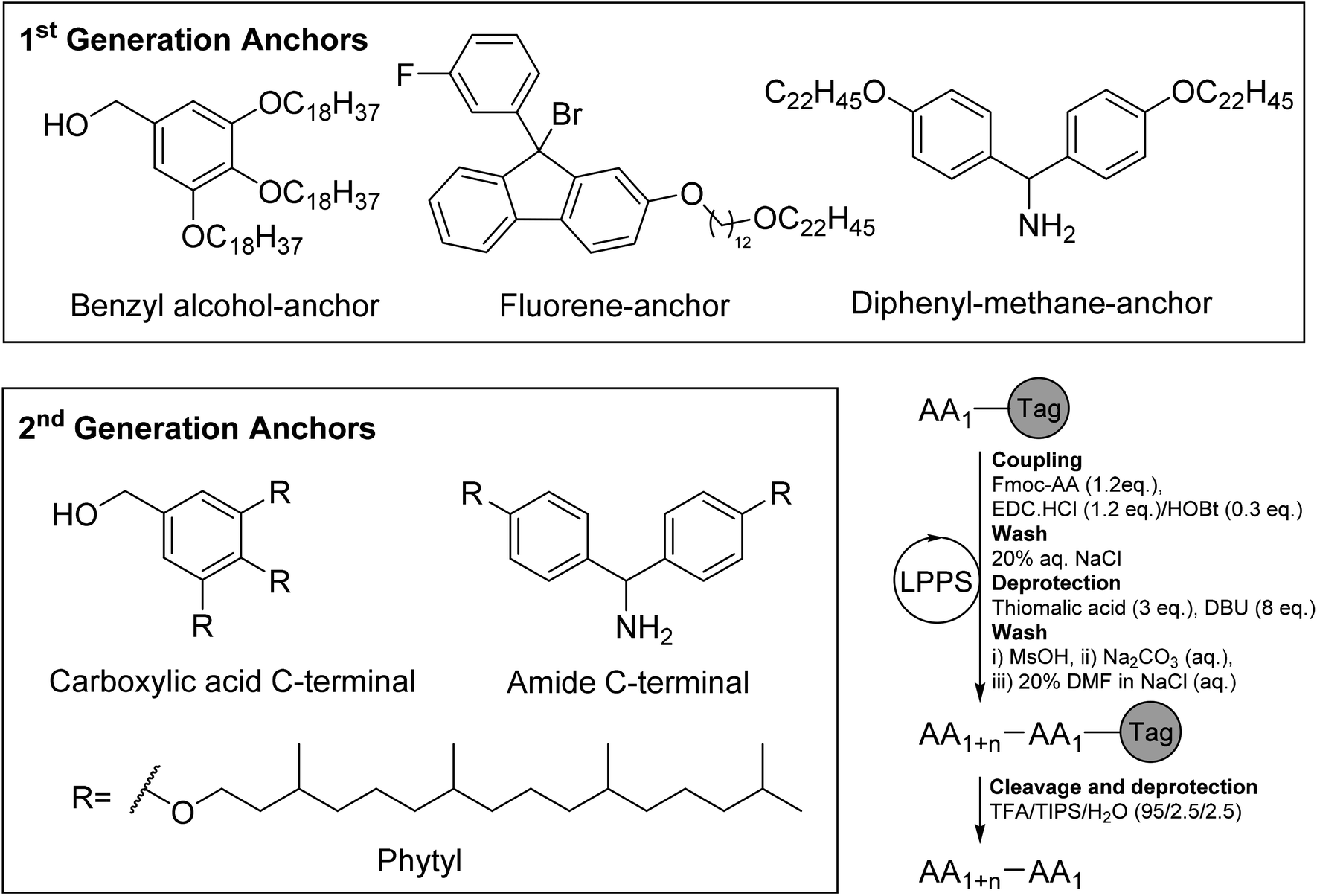
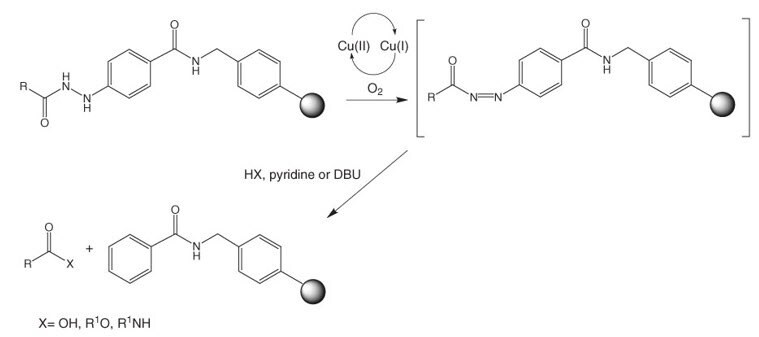
Greening the synthesis of peptide therapeutics: an industrial perspective - RSC Advances (RSC Publishing) DOI:10.1039/D0RA07204D

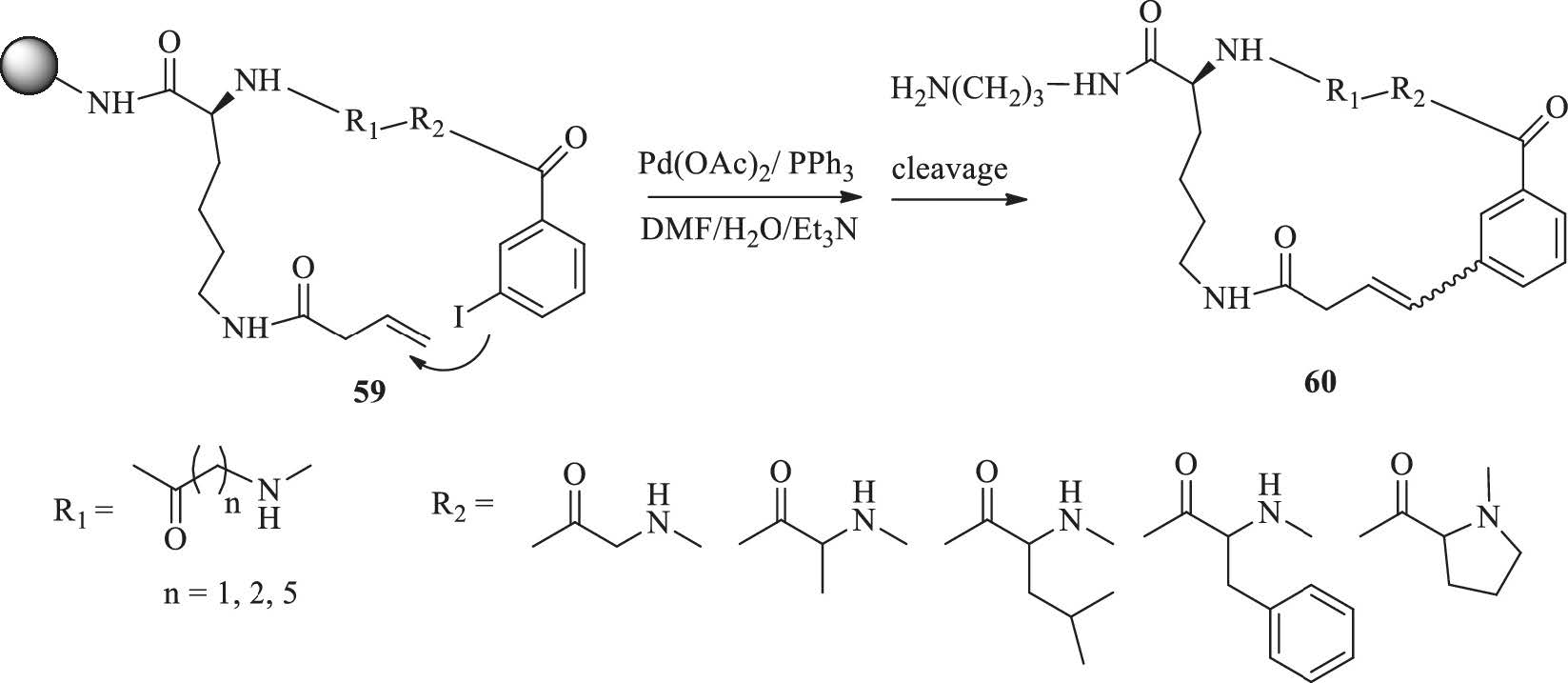
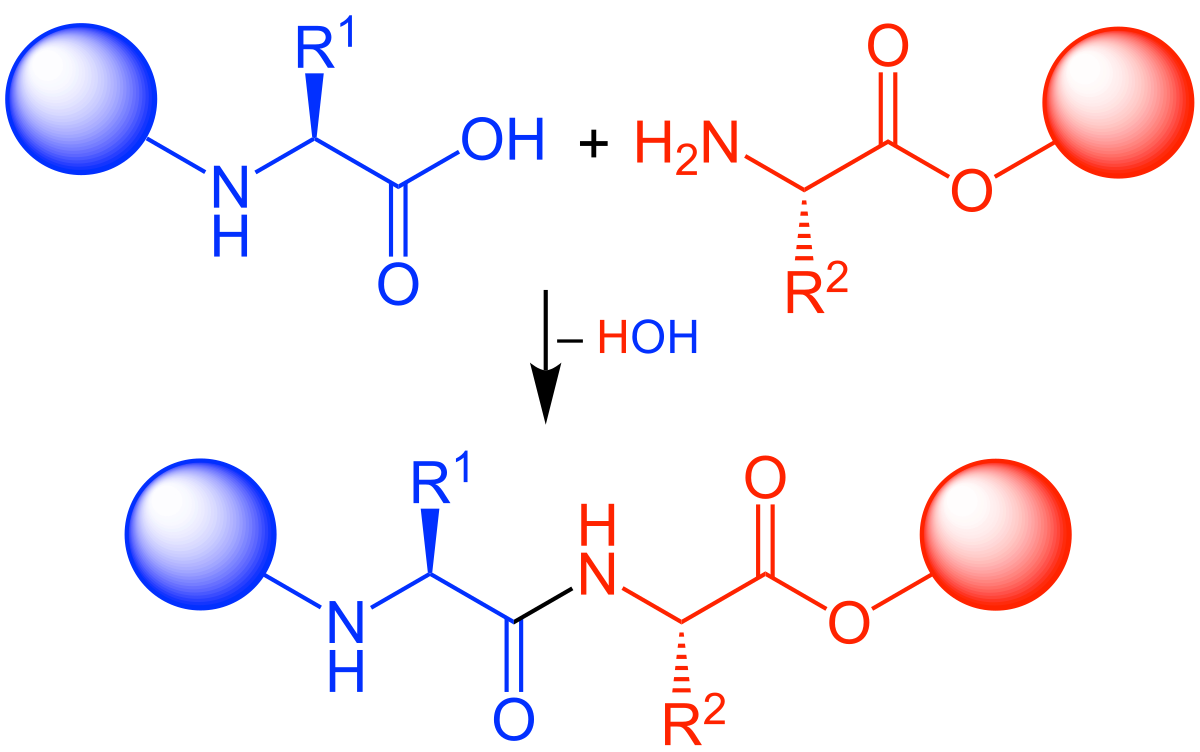
A general solid phase method for the synthesis of depsipeptides - Organic & Biomolecular Chemistry (RSC Publishing) DOI:10.1039/C2OB26893K