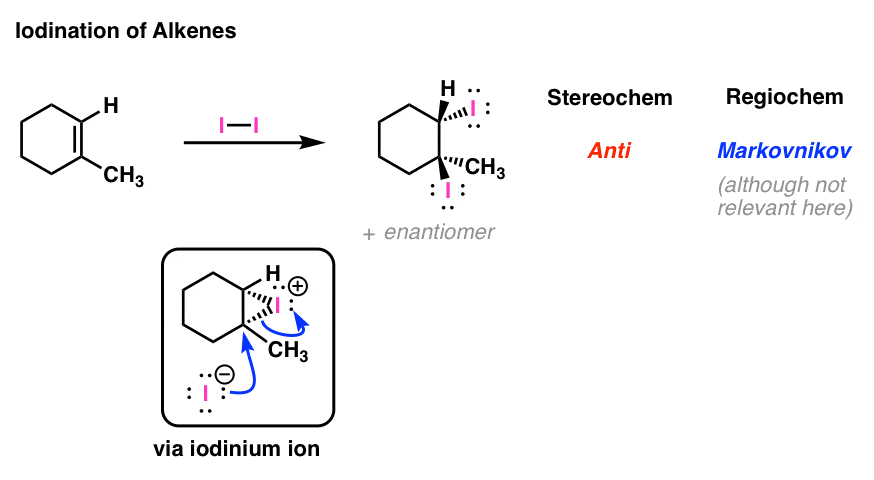
![PDF] Highly Regioselective Iodination of Arenes via Iron(III)-Catalyzed Activation of N-Iodosuccinimide. | Semantic Scholar PDF] Highly Regioselective Iodination of Arenes via Iron(III)-Catalyzed Activation of N-Iodosuccinimide. | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/0c6f0edeacfadccc931009ae2c66114570d95279/2-Table2-1.png)
PDF] Highly Regioselective Iodination of Arenes via Iron(III)-Catalyzed Activation of N-Iodosuccinimide. | Semantic Scholar

N-Iodosuccinimide-Promoted Cascade Trifunctionalization of Alkynoates: Access to 1,1-Diiodoalkenes | Organic Letters

N‐Iodosuccinimide (NIS) in Direct Aromatic Iodination - Bergström - 2017 - European Journal of Organic Chemistry - Wiley Online Library
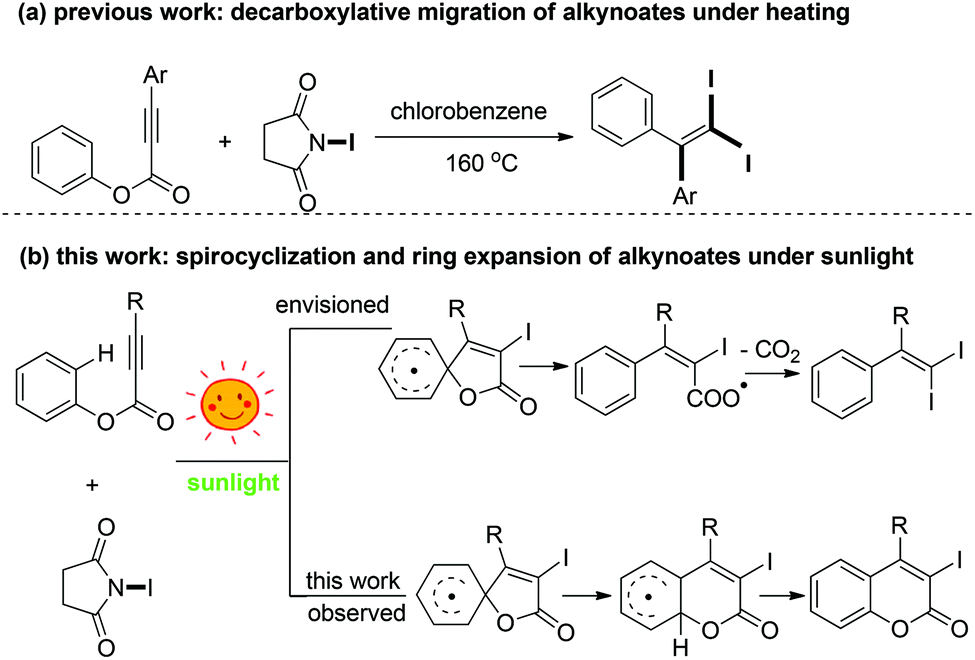
Sunlight-promoted cyclization versus decarboxylation in the reaction of alkynoates with N -iodosuccinimide: easy access to 3-iodocoumarins - Green Chemistry (RSC Publishing) DOI:10.1039/C6GC01027J
![PDF] Mild and regioselective iodination of electron-rich aromatics with N- iodosuccinimide and catalytic trifluoroacetic acid | Semantic Scholar PDF] Mild and regioselective iodination of electron-rich aromatics with N- iodosuccinimide and catalytic trifluoroacetic acid | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/857946e16a0a13e2aebbb7814400dc8f6bc9a574/2-Table1-1.png)
PDF] Mild and regioselective iodination of electron-rich aromatics with N- iodosuccinimide and catalytic trifluoroacetic acid | Semantic Scholar
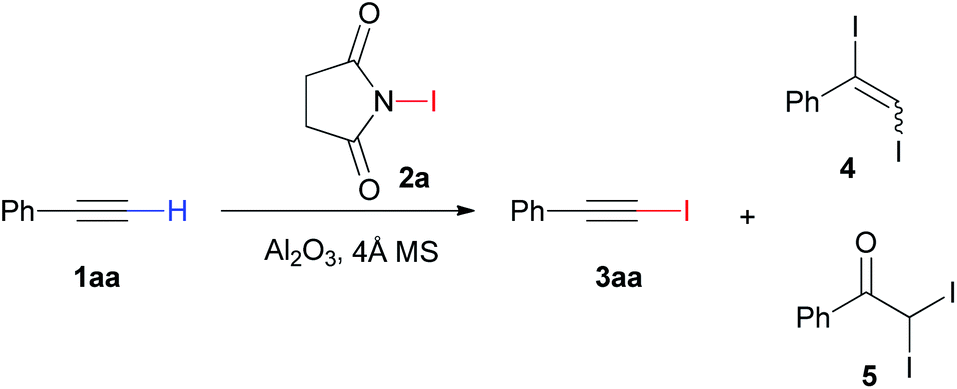
Efficient synthesis of 1-iodoalkynes via Al 2 O 3 mediated reaction of terminal alkynes and N -iodosuccinimide - RSC Advances (RSC Publishing) DOI:10.1039/D0RA00251H

Photoinduced cyclization of alkynoates to coumarins with N-Iodosuccinimide as a free-radical initiator under ambient and metal-free conditions - ScienceDirect

N‐Iodosuccinimide as Bifunctional Reagent in (E)‐Selective C(sp2)−H Sulfonylation of Styrenes - Pramanik - 2019 - Asian Journal of Organic Chemistry - Wiley Online Library

Scandium triflate-catalyzed 6,6′-diiodination of 2,2′-dimethoxy-1,1′-binaphthyl with 1,3-diiodo-5,5-dimethylhydantoin - ScienceDirect


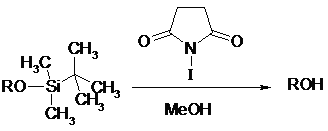

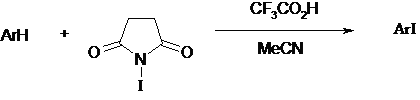

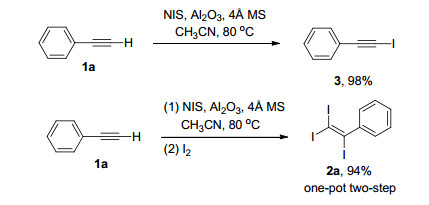



![Chemoselective iodination of 6-substituted imidazo[1,2-a]pyridine | SpringerLink Chemoselective iodination of 6-substituted imidazo[1,2-a]pyridine | SpringerLink](https://media.springernature.com/lw685/springer-static/image/art%3A10.1007%2Fs10593-018-2307-x/MediaObjects/10593_2018_2307_Figa_HTML.gif)