Cancers | Free Full-Text | FDA-Approved Drugs for Hematological Malignancies—The Last Decade Review | HTML
Cancers | Free Full-Text | FDA-Approved Drugs for Hematological Malignancies—The Last Decade Review | HTML
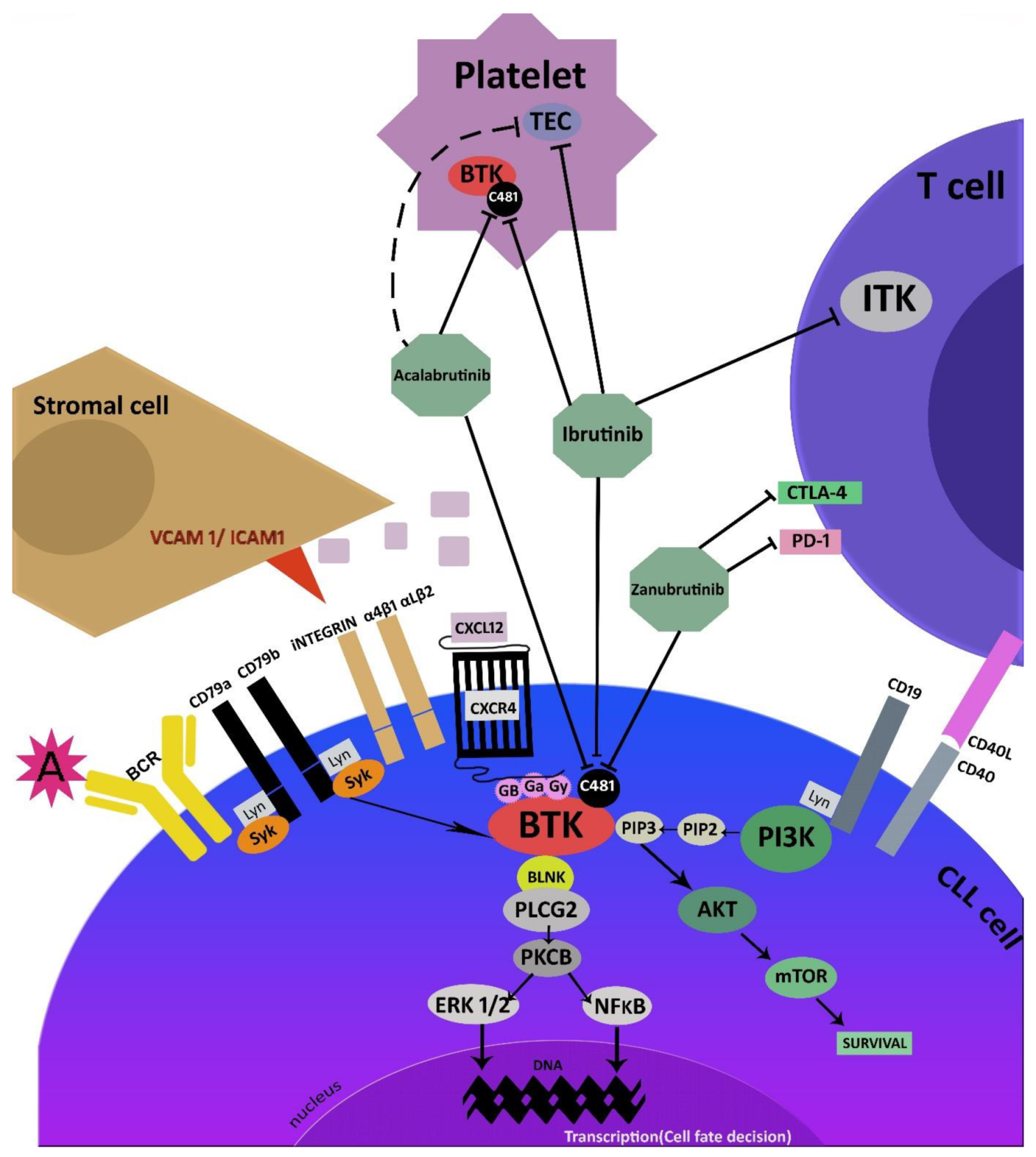
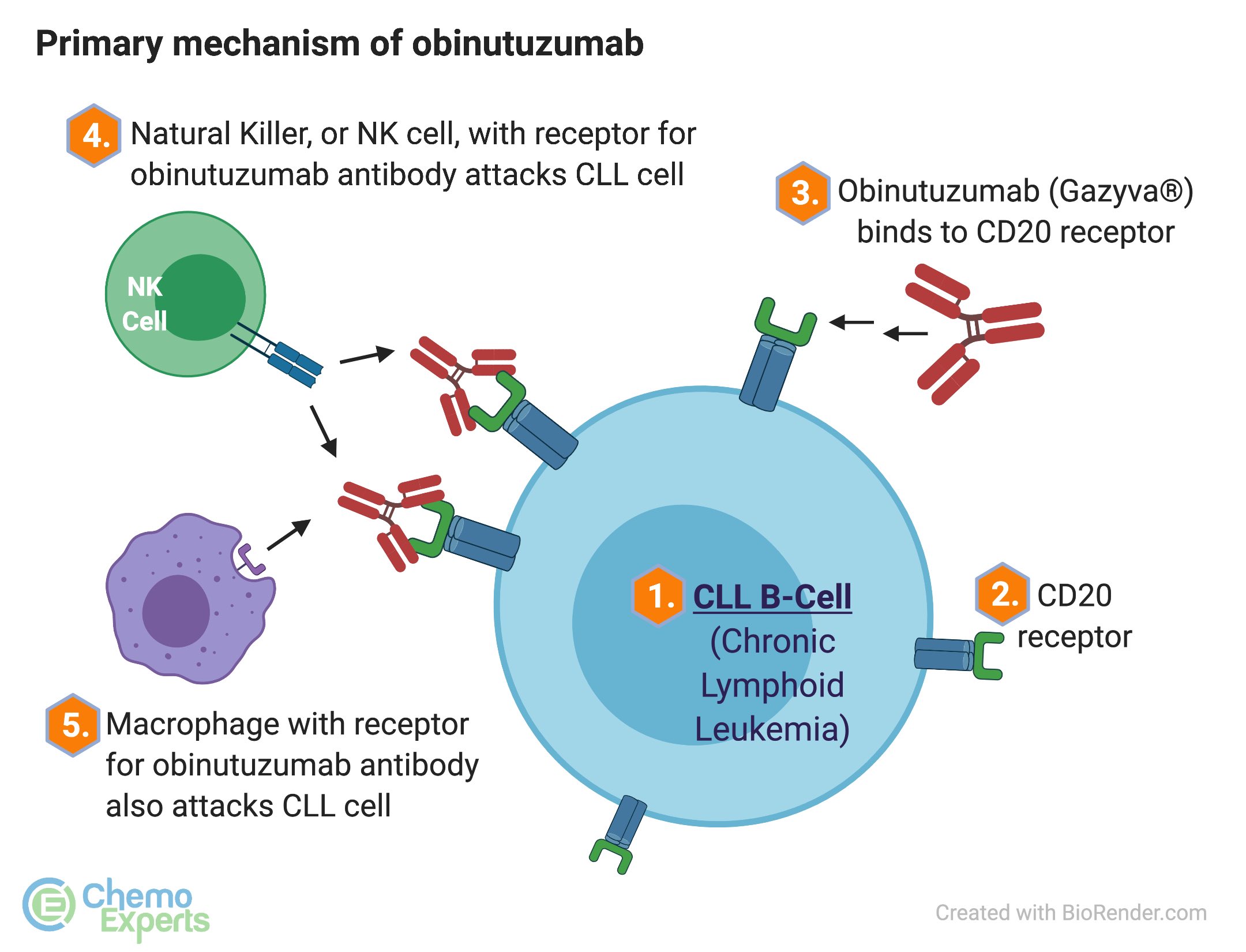
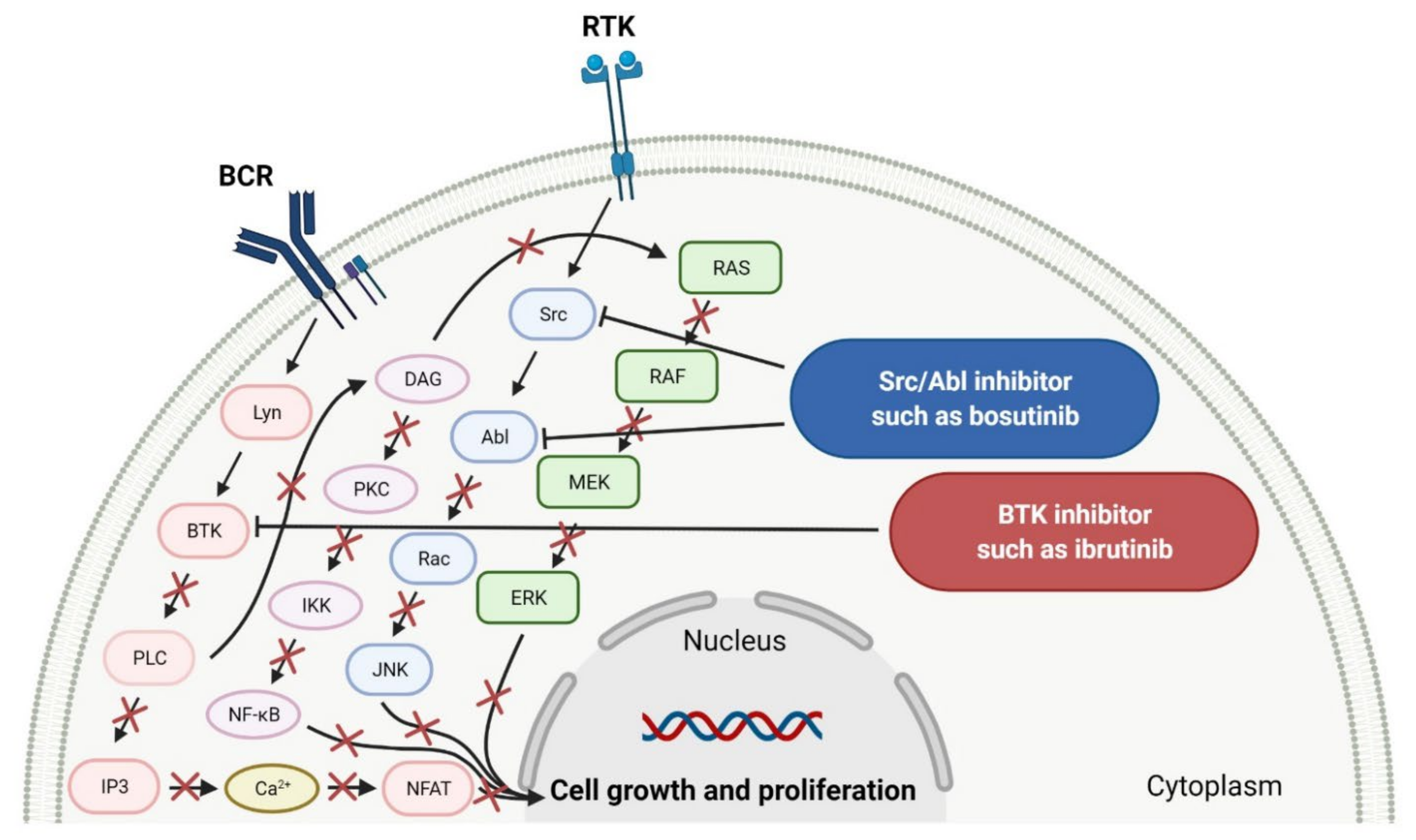
Cancers | Free Full-Text | The Role of Bruton’s Kinase Inhibitors in Chronic Lymphocytic Leukemia: Current Status and Future Directions | HTML

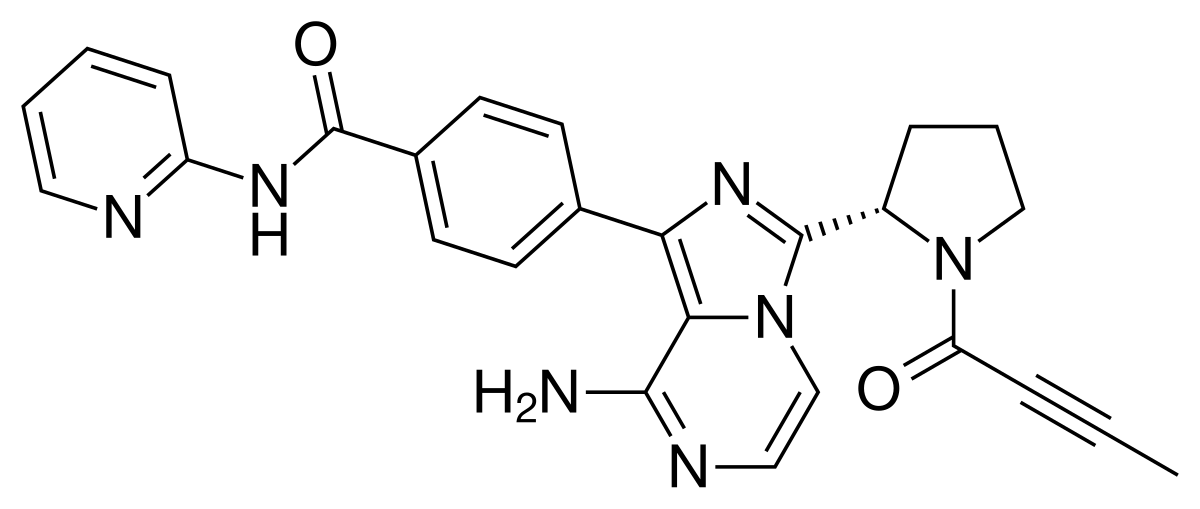
Bioavailability, Biotransformation, and Excretion of the Covalent Bruton Tyrosine Kinase Inhibitor Acalabrutinib in Rats, Dogs, and Humans | Drug Metabolism & Disposition

Balancing Potential Benefits and Risks of Bruton Tyrosine Kinase Inhibitor Therapies in Multiple Sclerosis During the COVID-19 Pandemic | Neurology Neuroimmunology & Neuroinflammation

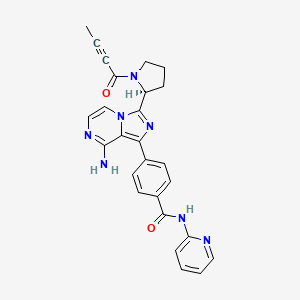
These highlights do not include all the information needed to use CALQUENCE safely and effectively. See full prescribing information for CALQUENCE. CALQUENCE® (acalabrutinib) capsules, for oral useInitial U.S. Approval: 2017
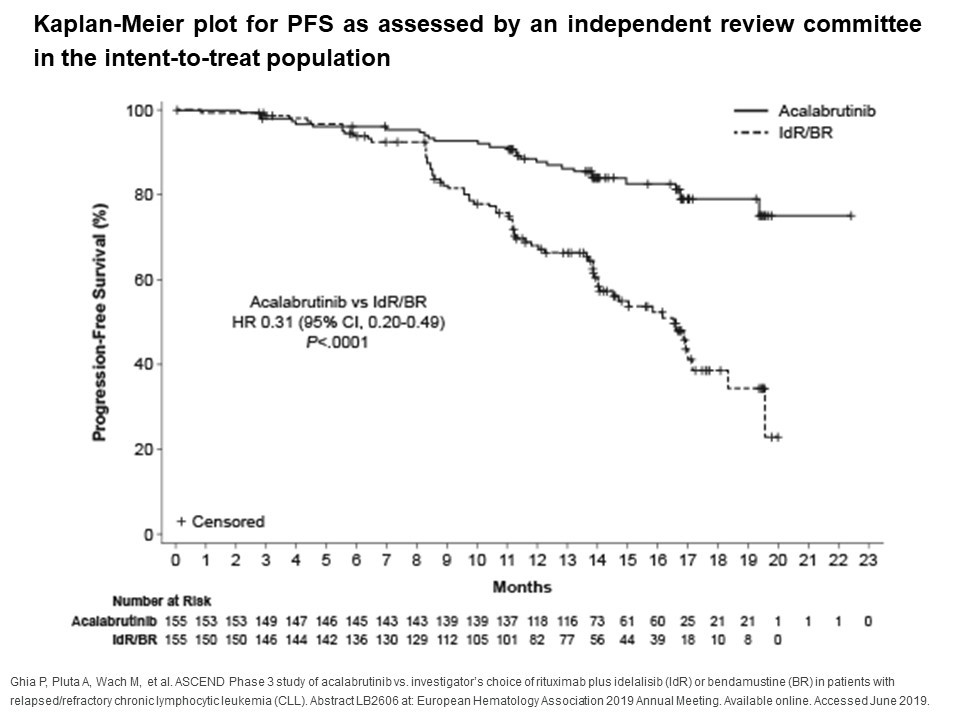
CALQUENCE® (acalabrutinib) Significantly Prolonged the Time Patients Lived without Disease Progression in Relapsed or Refractory Chronic Lymphocytic Leukemia | Business Wire

Bioavailability, Biotransformation, and Excretion of the Covalent Bruton Tyrosine Kinase Inhibitor Acalabrutinib in Rats, Dogs, and Humans | Drug Metabolism & Disposition

These highlights do not include all the information needed to use CALQUENCE safely and effectively. See full prescribing information for CALQUENCE. CALQUENCE® (acalabrutinib) capsules, for oral useInitial U.S. Approval: 2017